Catalytic, enantioselective annulations with chiral N-heterocyclic carbenes
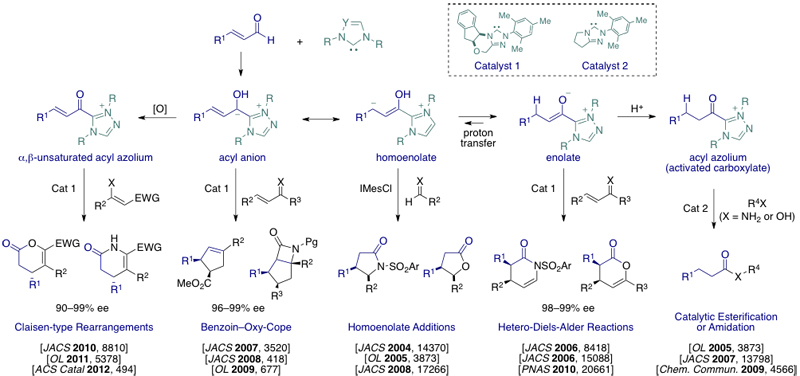
Previous efforts from our group led to an new branch of catalytic asymmetric synthesis, commonly known as “Chiral N-heterocyclic carbene (NHC) catalysis”. We pioneered the catalytic generation of new reactive species by the combination of N-heterocyclic carbenes and α-functionalized aldehydes. Since these first reports, we have expanded the NHC-catalyzed generation of reactive intermediates to the formation of four novel and distinct reactive intermediates: i) homoenolate equivalents, ii) enolate equivalents, iii) acyl azoliums, which serve as activated carboxylic acids, and iv) α,β-unsaturated acyl azoliums. Nearly all of these reactions are promoted by N-Mesityl substituted triazolium salts (1-2) developed in our group and currently sold by external page Aldrich, external page TCI, and external page Bioblocks. Examples from our group are shown below.
We are no longer pursuing NHC-catalyzed carbon–carbon bond formation in our group. Many other groups continue this research, which remains a fascinating and productive area of asymmetric catalysis.